题目
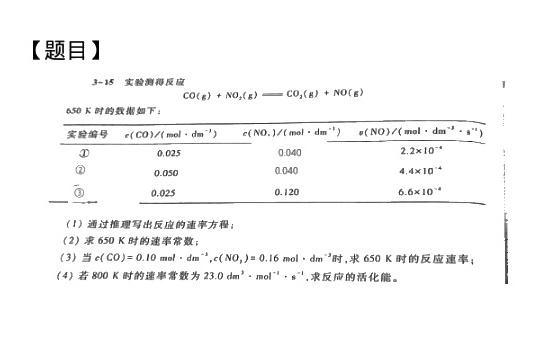
【题目】-|||-3-15 实验测得反应-|||-(g)+(N{O)_(2)}(g)=!=!= C(O)_(3)(g)+NO(g)-|||-650K时的数据如下:-|||-实验编号 (CO)ykparallel (molcdot (dm)^-3) ((Na)_(0))N(molcdot (dm)^-n) 哚哚 (NO)ykparallel (molcdot (dm)^-3cdot (s)^m)-|||-D 0.025 0.040 .2times (10)^-4-|||-② 0.050 0.040 .4times (10)^-4-|||-__ ā 0.025 0.120 .6times (10)^-4-|||-(1)通过推理写出反应的速率方程;-|||-(2)求650K时的速率常数;-|||-(3)当 (CO)=0.10molcdot (dm)^-2, (N(O)_(2))=0.16molcdot (dm)^-3 求650K时的反应速率;-|||-(4)若800K时的速率常数为 .0(dm)^3cdot mo(l)^+1cdot (m)^-1 求反应的活化能。
题目解答
答案
